Aaron Coleman
Research
Biology Education Research
All fields of biology are based on methodical experimentation and careful interpretation of data. Laboratory classes where students perform well-designed experiments using relevant techniques are an essential part of an undergraduate education in the biological sciences. An effective laboratory curriculum needs to immerse students in the experimental process. This means not only teaching the concepts underlying the laboratory work but also fostering the development of scientific reasoning. Our research investigates approaches to improve laboratory education on two fronts. First, we are trying to understand how students learn the scientific reasoning skills that are necessary for them to understand research and become scientifically-literate citizens. Second, we seek to design biochemistry laboratory modules that allow students to have a research-like experience in terms of the experimental systems and techniques used, but that are feasible to implement in high-enrollment courses.
Teaching Scientific Reasoning
The Vision and Change in Biology Undergraduate Education report (2011) from AAAS lists six core competencies that should be expected from graduates with a degree in the biological sciences. Competencies such as the ability to apply the process of science are best, if not exclusively, learned in the context of a laboratory class. Despite its importance, we are only beginning to understand how students learn the reasoning skills required to apply the process of science. In particular, what are the common misconceptions that interfere with this learning process, and what are the most effective strategies for teaching these skills in the laboratory? These questions are the focus of our research.
One area of investigation has been to determine how students use the concepts of correlation, necessity, and sufficiency in interpreting experimental data. These concepts are derived from the basic idea of correlation vs. causation, and using them correctly is essential for reading and evaluating the scientific literature. We found that students frequently make two specific types of reasoning errors that interfere with their ability to correctly interpret data (Coleman et al., 2015). When presented with an experiment that only establishes a correlation between a potential intermediate and a known effect, students frequently interpret the intermediate as being sufficient (causative) for the effect. Also, when presented with an experiment that only determines the necessity of an intermediate for an effect, students frequently interpret it as also providing information on the sufficiency of the intermediate, and vice versa. By identifying these common errors, lab course curricula can be modified to correct for them and more successfully teach these scientific reasoning skills.
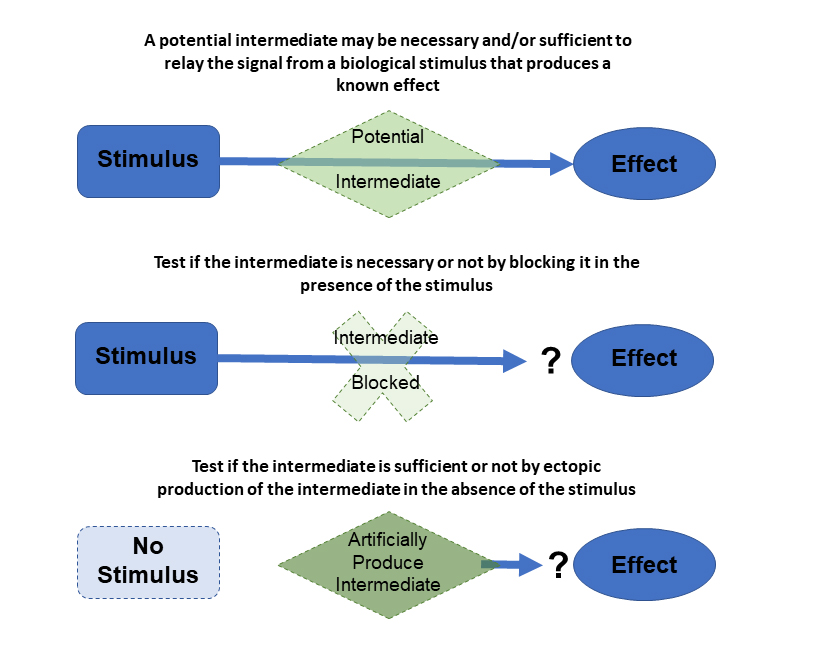
Another ongoing area of investigation is to understand how the degree of inquiry in a laboratory course affects the learning of scientific reasoning skills. Inquiry is defined as the students determining one or more elements of the scientific process carried out in a lab experiment or project, rather than these being provided by the instructor. We have created a rubric of inquiry elements, such as interpreting preliminary data to form a hypothesis or designing an experiment by selecting different experimental conditions to test. Allowing the students to determine these elements is thought to improve learning outcomes but increases the logistical difficulty of implementing the experiment, particularly in a high-enrollment course. By understanding which inquiry-elements are most important for the learning of specific scientific reasoning skills, these competing factors can be balanced to maximize learning outcomes within the constraints of a given lab course. We are currently studying how specific inquiry elements affect students’ learning of hypothesis formation, data interpretation, and experimental design.
Research on Fibroblast Growth Factor Signal Transduction
There are 18 members of the fibroblast growth factor (FGF) family that act in a paracrine or endocrine fashion to accomplish a diverse array of biological effects, from pattern formation during development to wound healing. All of this is mediated through only four transmembrane FGF receptors. I am interested in deciphering the signal transduction by which individual FGFs produce their unique biological responses.
Select Publications
- Kram, K.E., Yim, K.M., Coleman A.B. and Sato, B.K. (2016) Battle of the bacteria: Characterizing the evolutionary advantage of stationary phase growth. J. Microbiology and Biology Education 17:261-268.
- Coleman, A.B., Lam, D.P., Soowal, L.N. (2015) Correlation, Necessity, and Sufficiency: Common Errors in the Scientific Reasoning of Undergraduate Students for Interpreting Experiments. Biochemistry and Molecular Biology Education 43:305 – 315.
- Coleman, A.B. (2010) New Ideas for an Old Enzyme: A short, question-based laboratory project for the purification and identification of an unknown LDH isozyme.
- Biochemistry and Molecular Biology Education 38:253-260.
- Coleman, A.B. (2003). Positive and negative regulation of cellular sensitivity to anti-cancer drugs by FGF-2. Drug Resistance Updates6:85-94.
- Coleman, A.B., Metz, M., Donohue, C., Schwarz, R., and Kane, S.E. (2002) Chemosensitization by fibroblast growth factor-2 is not dependent on proliferation, S-phase accumulation, or p53 status. Biochemical Pharmacology64:1111-1123.
- Coleman, A.B., Momand, J., and Kane, S.E. (2000) Basic fibroblast growth factor sensitizes NIH 3T3 cells to apoptosis induced by cisplatin. Molecular Pharmacology 57:324-333.
Biography
Aaron Coleman obtained his Ph.D. in Biomedical Sciences from the University of California, Riverside. He conducted post-doctoral research at the Beckman Research Institute at City of Hope National Medical Center. He joined the Division of Biological Sciences in 2007.

