Alexandra Jazz Dickinson
Research
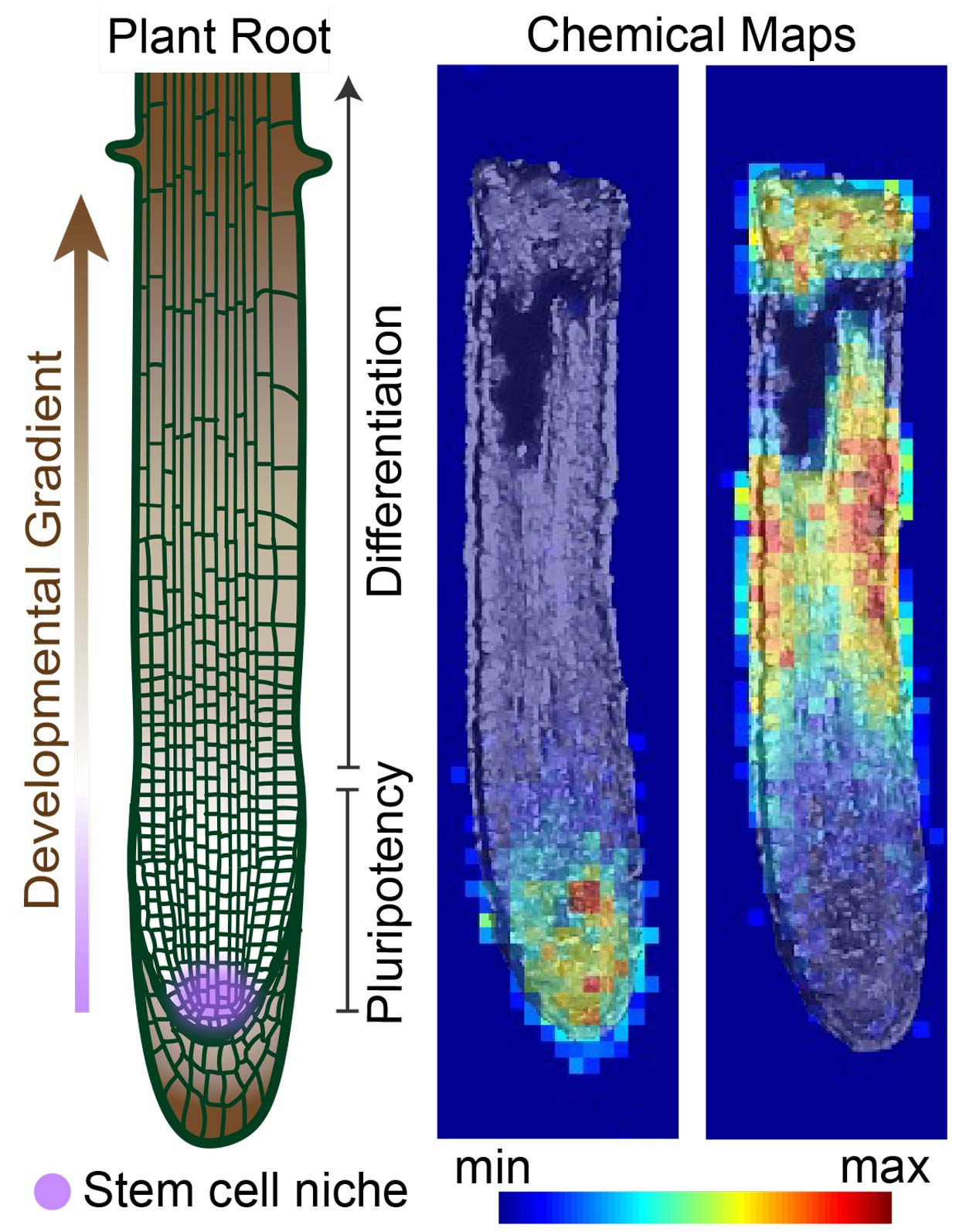 Figure 1. Chemical imaging using desorption electrospray ionization (DESI) mass spectrometry has the potential to unlock new developmental regulators.
Figure 1. Chemical imaging using desorption electrospray ionization (DESI) mass spectrometry has the potential to unlock new developmental regulators.Organ development requires regulated spatial and temporal coordination of stem cell division, tissue differentiation, and cellular maintenance. Accordingly, dysregulation of this balance can lead to defects in cell behavior, tissue morphogenesis, and organ function. How stem cell behaviors are coordinated during morphogenesis remains a central question in developmental biology. Due to their cell-to-cell mobility, small signaling molecules such as metabolites and lipids are instrumental for multicellular development. However, due to the challenges of identifying and perturbing metabolites in vivo, the specific functions of signaling metabolites in stem cell biology have remained largely uncharacterized. The principal goal of my research program is to identify metabolites that regulate stem cell function and determine their mechanisms of action.
Specifically, my lab applies high spatial resolution metabolomics, chemical genetics, and molecular biology to uncover novel signaling pathways in the Arabidopsis root meristem. We utilize the Arabidopsis root because the full developmental trajectory of stem cells is optically accessible, genetically tractable, and amenable to high-throughput phenotyping. Plants are also abundant producers of diverse metabolites, generating a broad parameter space to investigate the range of metabolite-regulated cellular processes. Importantly, molecular principles governing metabolite control of stem cell behavior can be conserved between plants and animals, with implications for applications ranging from agriculture to regenerative medicine. This work has led to the discovery of a number of new signaling molecules that regulate stem cell decisions.
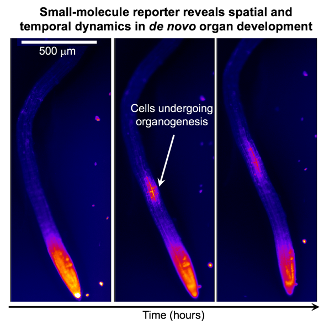 Figure 2. A fluorescent reporter reveals sites of organogenesis in the differentiating regions of Arabidopsis roots.
Figure 2. A fluorescent reporter reveals sites of organogenesis in the differentiating regions of Arabidopsis roots.Select Publications
- Dickinson, A. J., Zhang, J., Luciano, M., Wachsman, G., Sandoval, E., Schnermann, M., Dinneny, J. R., & Benfey, P. N. “A plant lipocalin promotes retinal-mediated oscillatory lateral root initiation.” Science, 373, 2021.
- Dickinson, A. J., Lehner, K., Mi, J., Jia, K., Mijar, M., Dinneny, J., Al-Babili, S., Benfey, P. N. “Beta-cyclocitral is a conserved root growth regulator.” PNAS, 116, 2019.
- Jia, K.,Dickinson, A. J., Mi, J., Cui, G., Kharbatia, N. M., Guo, X., Sugiono, E., Aranda, M., Rueping, M., Benfey, P. N., Al-Babili, S., “Anchorene is an endogenous diapocarotenoid required for anchor root formation in Arabidopsis.” Science Advances, 5, 2019.
- Dickinson, A. J., Meyer, M., Pawlak E. A., Gomez, S. M., Jaspers, I., Allbritton, N. L. “Analysis of the sphingosine kinase pathway in single natural killer cells enriched from peripheral blood.” Integrative Biology 2015, 4, 392-401 (cover).
- Dickinson, A. J., Hunsucker, S. A., Armistead, P. M., Allbritton, N. L. “Single-cell sphingosine kinase activity measurements in primary leukemia.” Analytical and Bioanalytical Chemistry 2014, 406, 7027-7036.
- Kovarik, M. L., Dickinson, A. J., Roy, P., Poonnen, R. A., Fine, J. P., Allbritton, N. L. “Response of single leukemic cells to peptidase inhibitor therapy across time and dose using a microfluidic device.” Integrative Biology 2014, 6, 164-174.
- Dickinson, A. J., Armistead, P. M., Allbritton, N. L. “Automated capillary electrophoresis system for fast single-cell analysis.” Analytical Chemistry 2013, 85, 4797-4804.
- Kovarik, M. L., Ornoff, D. M., Melvin, A. T., Dobes, N. C., Wang, Y. L., Dickinson, A. J., Gach, P. C., Shah, P. K., Allbritton, N. L. “Micro total analysis systems: fundamental advances and applications in the laboratory, clinic, and field.” Analytical Chemistry 2013, 85, 451-472.
- McKitterick, C. B., Erb-Stullo, N. L., Dickinson, A. J., and Collings, P. J. “Aggregation Properties of Chromonic Liquid Crystal Benzopurpurin 4B.” Journal of Physical Chemistry B 2010, 114, 1888-1896.
- Collings, P.J., Dickinson, A. J., and Smith, E. C. “Molecular aggregation and chromonic liquid crystals.” Liquid Crystals 2010, 37, 701-710.
- Dickinson, A. J., LaRacuente, N. D., McKitterick, C. B., Collings, P. J. “Aggregate structure and free energy changes in chromonic liquid crystals.” Molecular Crystals and Liquid Crystals 2009, 509, 751-762.
Biography
Jazz Dickinson received her PhD in chemistry from University of North Carolina – Chapel Hill. She conducted her post-doctoral research in developmental plant biology with Prof. Philip Benfey at Duke University. She has also served as a visiting scientist at Stanford University and is a recipient of the Arnold O. Beckman Postdoctoral Fellowship Award, the Boka W. Hadzija Award for Distinguished University Service at UNC-CH, and the Ruth L. Kirschstein Graduate Fellowship Award. She joined the UCSD faculty in 2020 to pursue research in small molecule regulation of development.

