Byungkook Lim
Research
Lab of Functional Anatomy of the Neural Circuitry
The basal ganglia and interconnected cortical, amygdalar and thalamic circuits subserve several crucial brain functions including reward-based learning, motor learning, motivated behavior and stress-related behaviors. Dysfunction in these circuits due to alterations of synaptic properties has been proposed to play a critical role in a variety of neuropsychiatric disorders including anxiety, addiction, autism spectrum disorder and major depressive disorder. However, the brain circuits relaying information from or to basal ganglia and their detailed synaptic organization have not been clearly delineated.
The main objective of research in our laboratory is to understand the role of the circuit-specific synaptic organization in basal ganglia in animal models of neuropsychiatric disease including drug addiction, feeding disorder, depressive disorder, and movement disorder. Given the circuit and synaptic complexity of the brain, characterizing specific neural circuitry responsible for behavioral changes has been challenging.
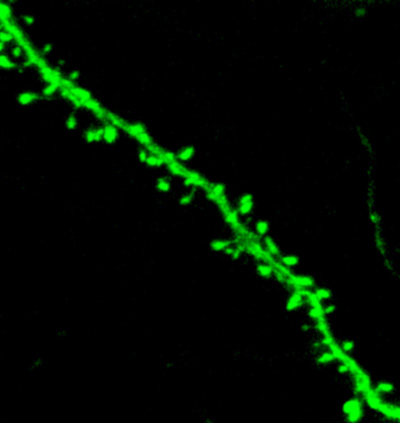
Dendtiric spines of amygdala neurons projecting to dopamine receptor 1-expressing medium spiny neurons (D1-MSNs) in nucleus accumbens
To address the anatomical and functional properties of basal ganglia circuitry, our laboratory applies and develops a variety of molecular strategies (ex. virus-mediated tracing and gene expression, transgenic mice expressing marker proteins in specific population of neurons) together with electrophysiological approaches. Furthermore, we applies cell-type specific or input-specific optogenetic manipulation in vitro and in vivo in pharmacological animal models of human disease as well as a battery of behavioral testing.
Future research in my own lab will continue with this strategy and set out to anatomically and functionally define neuronal identity and connectivity of brain circuits beyond basal ganglia and their interplay in mediating reward- and motivation-related behavior in the pathogenesis of neuropsychiatric and neuropathological disease.
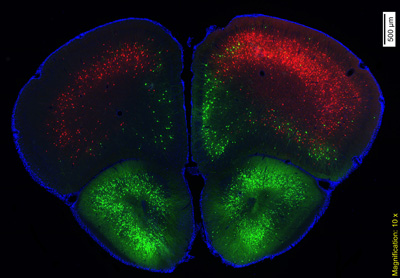
Double rabies virus-mediated retrograde tracing showing cortical neurons projecting to dorsal striatum (red) and neurons projecting to nucleus accumbens (green).
Select Publications
- Shin, S., Pribiag, H., Lilascharoen, V., Knowland, D., Wang, X., Lim, B. K. (2018) Drd3 signaling in the lateral septum mediates early life stress-induced social dysfunction. Neuron, 2018 Jan 3.
- Liu, J., Conde, K., Zhang, P., Lilascharoen, V., Xu, Z., Lim, B. K., Seeley, R. J., Zhu, J. J., Scott, M. M., Pang, Z.P. (2017) Enhanced AMPA Receptor Trafficking Mediates the Anorexigenic Effect of ndogenous Glucagon-like Peptide-1 in the Paraventricular Hypothalamus. Neuron. 2017 Oct 13.
- Knowland, D., Lilascharoen, V., Pacia, C. C., Shin, D., Wang, E., Lim, B. K. (2017) Distinct ventral pallidal neural populations mediate separate symptoms of depression. Cell Jul 13;170(2):284-297
- Yoo, J. H., Zell, V., Wu, J., Punta, C., Ramajayam, N., Shen, X., Faget, L., Lilascharoen, V., Lim, B. K., Hnasko, T. S. (2017) Activation of Pedunculopontine Glutamate Neurons Is Reinforcing. J Neurosci. 37(1):38-46.
- Rothwell, P. E., Hayton, S. J., Sun, G. L., Fuccillo, M. V., Lim, B. K., Malenka, R. C. (2015) Input- and Output-Specific Regulation of Serial Order Performance by Corticostriatal Circuits. Neuron 88(2):345-356.
- Schwartz, N., Temkin, P., Jurado, S., Lim, B. K., Polepalli, J. S., Malenka, R. C. (2014) Decreased motivation during chronic pain requires plasticity in the nucleus accumbens core. Science 345(6196) 535-542.
- Rothwell, P. E, Fuccillo, M. V., Maxeiner, S., Hayton, S. J., Lim, B. K., Malenka, R. C., Südhof, T. C. (2014) Autism-associated Neuroligin-3 mutations impair striatal circuits underlying repetitive motor routins. Cell. 158(1):198-212.
- Lammel, S.*, Lim, B. K.*, Malenka, R. C. (2014) Reward and aversion in a heterogeneous midbrain dopamine system. Neuropharmacology. 76 Pt. B: 351-9 (* equal contribution)
- Kim, S. Y., Adhikari, A., Lee, S. Y., Marshel, J. H., Kim, C. K., Mallory, C. S., Lo, M., Pak, S., Mattis, J., Lim, B. K., Malenka, R. C., Warden, M. R., Neve, R., Tye, K. M., Deisseroth, K. (2013) Diverging neural pathways assemble a behavioural state from separable features in anxiety. Nature. 496(7444): 219-223.
- Lammel, S.*, Lim, B. K.*, Ran, C., Huang, K. W., Tye, K., Deisseroth, K., Malenka, R. C. (2012) Input-specific control of reward and aversion in the ventral tegmental area. Nature (Article) 491(7423): 212-217 (* equal contribution)
- Lim, B. K., Huang, K. W., Grueter, B. A., Rothwell, P. E., Malenka, R. C. (2012) Anhedonia requires MC4R-mediated synaptic adaptation in nucleus accumbens. Nature (Article) 487(7406): 183-189.
- Shelly, M., Cancedda, L., Lim, B. K., Popescu, A. T., Cheng, P. L., Gao, H., Poo, M. M. (2011) Semaphorin3A regulates neuronal polarization by suppressing axon formation and promoting dendrite growth. Neuron. 71(3):433-446.
- Lu, H., Cheng, P. L., Lim, B. K., Khoshnevisrad, N., Poo, M. M. (2010) Elevated BDNF expression after cocaine withdrawal facilitates LTP in medial prefrontal cortex by suppressing GABA inhibition. Neuron. 67(5):821-833.
- Lim, B. K., Cho, S. J., Sumbre, G., Poo, M. M. (2010) Region-specific contribution of ephrin-B and Wnt signaling to receptive field plasticity in developing optic tectum. Neuron. 65(6):899-911.
- Shelly, M.*, Lim, B. K.*, Cancedda, L., Heilshorn, S. C., Gao, H., Poo, M. M. (2010) Local and long-range reciprocal regulation of cAMP and cGMP in axon/dendrite formation. Science. (Article) 327(5965):547-552. (* equal contribution)
- Lu, H., Lim, B., Poo, M. M. (2009) Cocaine exposure in utero alters synaptic plasticity in the medial prefrontal cortex of postnatal rats. J Neurosci. 29(40):12664-12674.
- Lim, B. K., Matsuda, N., Poo, M. M. (2008) Ephrin-B reverse signaling promotes structural and functional synaptic maturation in vivo. Nat Neurosci. 11(2):160-169.
Biography
Byungkook Lim received his Ph.D. in Molecular & Cell Biology from UC Berkeley with Dr. Mu-ming Poo and carried out his postdoctoral training with Dr. Robert Malenka in the Department of Psychiatry and Behavioral Science at the Stanford University School of Medicine. Byungkook is a recipient of Kingenstein-Simmons Fellowship, Searle Scholar Fellowship, NIMH Biobehavioral Research Awards for Innovative New Scientists (BRAINS) and BRAIN Initiative Cell Census Network (BICCN) awards.

