Maurice Montal
Research
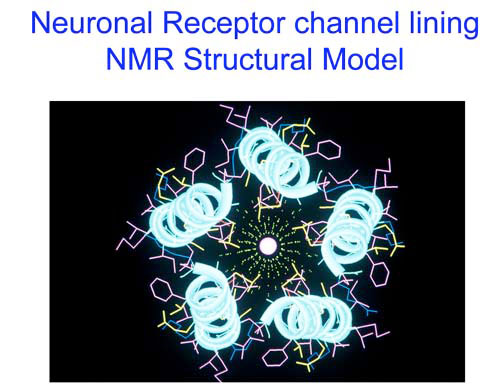
The key scientific question has been how membrane proteins function in terms of the underlying structure. These entities are truly smart materials. Proteins are nanomachines and marvels of protein chemistry. Lipid bilayers, the basic structural elements of biological membranes, are nanodevices which exploit self-assembly principles to provide the exquisite environment for proteins to operate. Together as a system, proteins embedded within lipid bilayers, embody the basis for biosensor and nanosensor technology. They also emerge as templates for molecular mimicry and design and follow the hierarchical organization underpinning complex systems. Our programs are of prime significance to health science and biodefense; they also comprise components of relevance to the pharmaceutical and cosmeceutical industries.
Specifically, our aim is to understand the structures and the mechanisms underlying nerve cell function and dysfunction. We are interested in the molecular basis of the intrinsic excitability of neurons, the communication of excitability between neurons, neuronal cell survival and death, the processes leading to neurodegeneration, and the genetic components in the pathogenesis of some forms of infant epilepsy. Given that our subjects of inquiry are ion-conducting and protein-conducting channels, key elements of the nerve cell, they embody drug targets; therefore, our programs encompass a component directed to screen or discover agents that impact these processes and, may in due turn, become clinically important drugs.
Botulinum neurotoxin (BoNT) is considered the most potent toxin; it is the causative agent of botulism, a neuroparalytic illness, and a major bioweapon without an antidote. A research program is directed towards this goal. Our objective is to understand the intricacies of intracellular trafficking of BoNT that may disclose novel pathways to escort, target, and insert it into membranes. This information may prove diagnostic in identifying unsuspected sites for intervention and plausible strategies for targeted drug delivery. BoNT is a modular nanomachine in which one of its modules -- the heavy chain channel -- operates as a specific protein translocating transmembrane chaperone for another of its component modules -- the light chain protease. We investigate the dynamics of protein-translocation in neuronal cells focusing on the interactions between the channel/chaperone and its cargo using an assay with single molecule sensitivity. This strategy led us to uncover an entirely unknown succession of intermediate states during translocation of the cargo which reveal transient protein-protein interactions occurring at distinct stages during protease translocation through the channel.
Another major area of interest concerns the design of biosensors based on lipid bilayers membranes containing a number of designed channel proteins that emulate those naturally occurring in the brain. Such devices are indeed nanomachines which may display unparalleled sensitivity in analyte detection, including but not limited to biological and environmental toxins, and also in sensing physical stimuli such as light, temperature, pressure or electric fields.
Select Publications
- Fischer, A. and Montal, M. Characterization of Clostridial botulinum neurotoxin channels in neuroblastoma cells. Neurotoxicity Res. 9:93-100 (2006).
- Santos, J.S., Lundby, A., Zazueta, C. and Montal, M. Molecular template for a voltage sensor in a novel K-plus channel. I. Identification and functional characterization of KvLm, a voltage-gated K-plus channel from Listeria monocytogenes. J. Gen. Physiol. 128:283-292 (2006).
- Lundby, A., Santos, J.S., Zazueta, C. and Montal, M. Molecular template for a voltage sensor in a novel K-plus channel. II. Conservation of a eukaryotic sensor fold in a prokaryotic K-plus channel. J. Gen. Physiol. 128:293-300 (2006).
- Fischer, A. and Montal, M. Single molecule detection of intermediates during Botulinum neurotoxin translocation across membranes. Proc Natl Acad Sci U S A 104: 10447-10452 (2007).
- Brunger, A.T., Breidenbach, M.A., Jin, R., Fischer, A., Santos, J.S., and Montal, M. Botulinum neurotoxin heavy chain belt as an intramolecular chaperone for the light chain. PLoS Pathogens 3:1191-1194 (2007).
- Fischer, A., and Montal, M. Crucial role of the disulfide bridge between Botulinum neurotoxin light and heavy chains in protease translocation across membranes. J. Biol. Chem. 282: 29604-29611 (2007).
- Fischer, A., Garcia-Rodriguez, C., Geren, I., Lou, J., Marks, J.D., Nakagawa, T., and Montal, M. Molecular architecture of Botulinum neurotoxin E revealed by single particle electron microscopy. J. Biol. Chem. 283: 3997-4003 (2008). doi/10.1074, published online on November 20, 2007.
- Fischer, A., Koriazova, L., Oblatt-Montal, M. and Montal, M. Botulinum Neurotoxin -- A Modular Nanomachine. In: Botulinum Toxin: Therapeutic Clinical Practice and Science. Edited by J.J. Jankovic, A. Albanese, M. Z. Atassi, J.O. Dolly, M. Hallett and N.H. Mayer. Elsevier Publ. (in press).
- Fischer, A., Mushrush, D., Lacy, D. B., and Montal, M. Botulinum neurotoxin devoid of receptor binding domain translocates active protease. PLoS Pathogens (in press).
- Montal, M. Translocation of Botulinum neurotoxin light chain protease by the heavy chain protein-conducting channel. Special Issue of Toxicon (in press).
- Santos, J., Grigoryev, S. M. and Montal, M. Molecular template for a voltage sensor in a novel K-plus channel. III. Functional reconstitution of a sensor-less pore module from a prokaryotic K-plus channe. J. Gen. Physiol. (December 2008, in press).
- Montal, M. Vpu Matchmakers as a Therapeutic Strategy for HIV infection. PLoS Pathogens (in press).
Biography
Mauricio Montal received his Ph.D. from the University of Pennsylvania and his M.D. from the National University of Mexico. He is the recipient of a K.C. Cole Award of the Biophysical Society for outstanding contributions in Membrane Biophysics and a Research Scientist Award from NIMH. Dr. Montal is a recipient of a Gugenheim Fellowship and a Fellow of the Biophysical Society.

