Wait for Me: Cell Biologists Decipher Signal that Ensures No Chromosome is Left Behind
Researchers use novel probe to witness how a ‘matchmaker’ molecule puts the brake on cell division until components are ready to be split
January 11, 2021
By Mario Aguilera
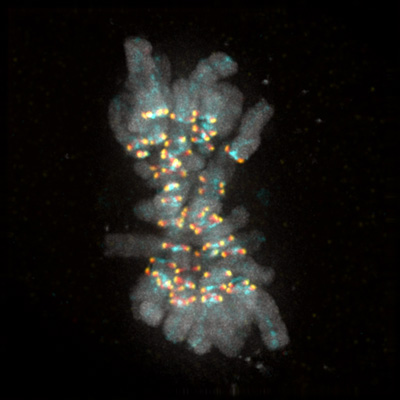
A human cell is featured during mitosis with its chromosomes (gray areas) aligned. The region known as the kinetochore (colored with yellow, red and cyan markers) is the origin of the checkpoint signal that acts as a safety check against premature cell division and is the focus of UC San Diego researchers’ study.
Starting as a single cell, organisms undergo millions of generations of divisions to ultimately generate the bones, heart, brain and other components that make up a living being. The mainspring within this intricate process is the transfer of DNA through each subsequent cell split within discrete packets called chromosomes.
It’s critical that all chromosomes are duplicated and precisely distributed through every generation of cell division. If the inherited chromosome components are altered, even slightly, birth defects and certain cancers can result.
A new study published in the journal Science by postdoctoral scholar Pablo Lara-Gonzalez, Division of Biological Sciences Professor Arshad Desai and their colleagues addresses the mystery of how chromosomes are inherited correctly every time a cell divides. Using a novel probe that monitors a key aspect of this process, Lara-Gonzalez and Desai have detailed the mechanics behind a “wait” signal that ensures that cell division isn’t prematurely set in motion.
The researchers concentrated their investigations on a pathway in the cell called the “spindle checkpoint,” which is a type of quality control mechanism that ensures accurate chromosome inheritance during cell division. The spindle checkpoint pathway is activated at a site on the chromosome called the kinetochore, a mechanical interface where protein fibers are coupled to pull chromosomes apart.
“When kinetochores are not attached to these protein fibers, they send out a ‘wait’ signal that halts the cell in mitosis (cell division), thereby giving time for attachments to be formed,” said Desai, a professor in the Section of Cell and Developmental Biology (Biological Sciences) and the Department of Cellular and Molecular Medicine (School of Medicine). “In this way, the cell makes sure all chromosomes are attached properly and ready to be pulled apart before the cell divides, thereby leaving no chromosome behind.”
In the Science paper, the researchers describe how the wait checkpoint signal is specifically generated at kinetochores of unattached chromosomes. Serendipitously, they developed a fluorescent probe that enabled them to watch for the first time the key molecular event in wait signal generation at kinetochores in living cells.
“This work identified a key ‘matchmaker’ molecule that brings together two constituents of the wait signal that do not like to associate with each other on their own,” said Lara-Gonzalez. “These findings help explain why the ‘wait’ checkpoint signal is selectively generated at kinetochores and not elsewhere in the cell.”
The findings offer a framework for ways in which the accuracy of chromosome inheritance can be lowered in certain states of disease, such as cancer, the researchers said.
Coauthors of the paper are: Pablo Lara-Gonzalez, Taekyung Kim, Karen Oegema, Kevin Corbett and Arshad Desai.
Funding for the research was provided by the National Institutes of Health (R01GM074215, R01GM104141); Ludwig Institute for Cancer Research; S10 OD021724 (UC San Diego mass spectrometry); and the National Research Foundation of Korea (2020R1C1C1008696).
