Hiruy Meharena
Research
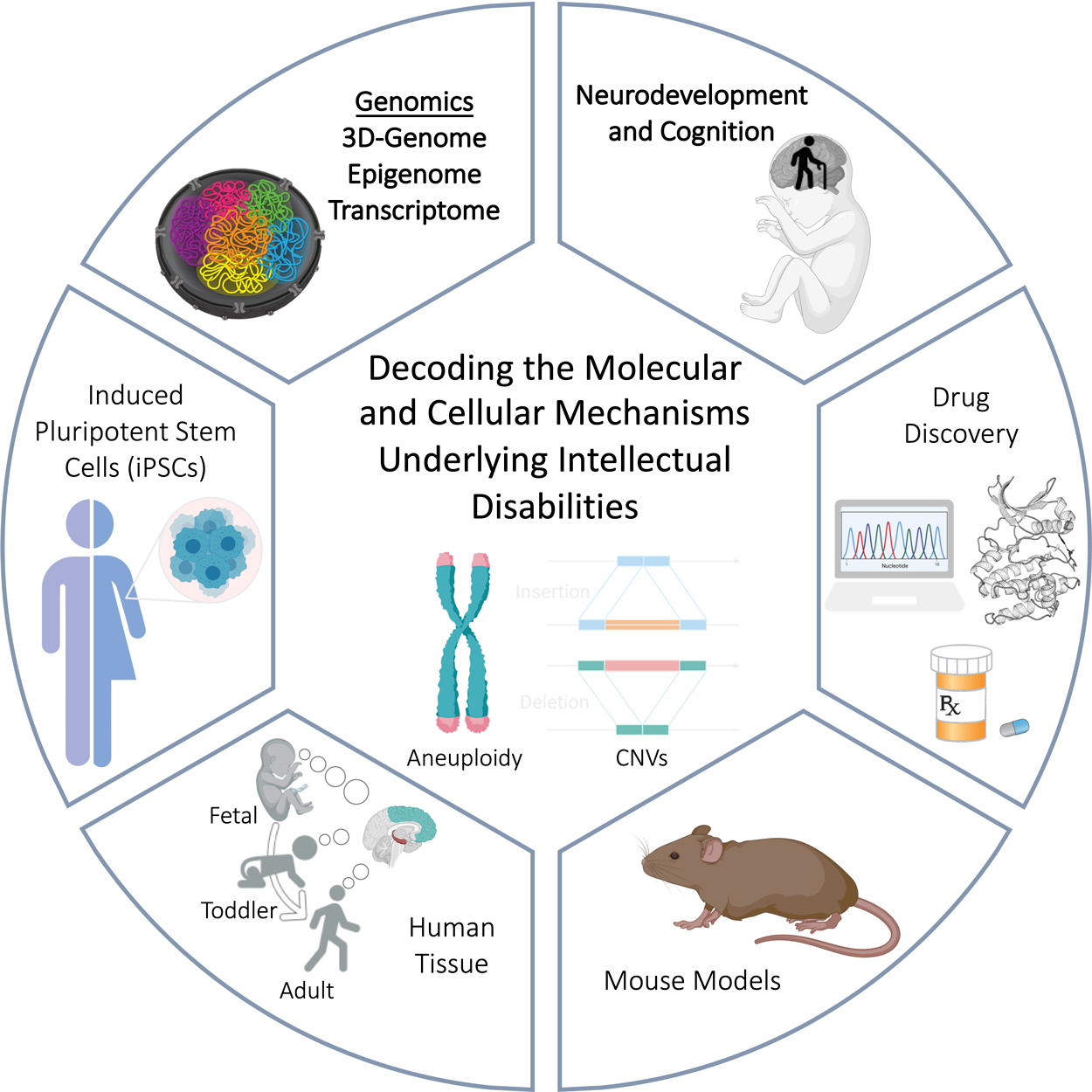
The overarching goal of our group is to establish a comprehensive molecular map of the developing human brain in order to identify the cell-type specific gene-networks driving neurodevelopmental disorders associated with intellectual disability, including Autism Spectrum Disorder and Down syndrome. We utilize mouse models and human patient derived induced pluripotent stem cells (iPSCs) to decode the genomic and molecular signatures of memory and learning and to identify how these processes are disrupted in neurodevelopmental disorders. Through collaborations with scientists, clinicians, and families we hope to identify new therapeutic avenues for individuals with intellectual disabilities.
Select Publications
- Meharena HS, Marco A, Dileep V, Lockshin ER, Kuffner G, Mullahoo J, Watson LA, Ko T, Guerin L, Abdurob F, Rengarajan S, Papanastasiou M, Jaffe JD, Tsai L-H. Down Syndrome Induced Senescence Disrupts the Nuclear Architecture of Neural Progenitors. ( In-press Cell Stem Cell)
- Marco, A., Meharena, H.S., Dileep, V. et al. Mapping the epigenomic and transcriptomic interplay during memory formation and recall in the hippocampal engram ensemble. Nat Neurosci (2020). https://doi.org/10.1038/s41593-020-00717-0
- Meharena, H.S., Fan, X., Ahuja, L.G., Keshwani, M.M., McClendon, C.L., N., Adams, J.A., and Taylor, S.S. (2016). Decoding the Interactions Regulating the Active State Mechanics of Eukaryotic Protein Kinases. PLoS Biol. 2016 Nov 30;14(11):e2000127
- Hu, J., Ahuja, L.G., Meharena, H.S., Kannan, N., Kornev, A.P., Taylor, S.S., and Shaw, A.S. (2015). Kinase regulation by hydrophobic spine assembly in cancer. Mol Cell Biol 35, 264-276.
- Pence, M.A., Haste, N.M., Meharena, H.S., Olson, J., Gallo, R.L., Nizet, V., and Kristian, S.A. (2015). Beta-Lactamase Repressor BlaI Modulates Staphylococcus aureus Cathelicidin Antimicrobial Peptide Resistance and Virulence. PloS one 10, e0136605.
- Zan, J., Choi, O., Meharena, H.M., Uhlson, C.L., Churchill, M.E., Hill, R.T., and Fuqua, C. (2015). A solo luxI-type gene directs acylhomoserine lactone synthesis and contributes to motility control in the marine sponge symbiont Ruegeria sp. KLH11. Microbiology 161, 50-56.
- Meharena, H.S., Chang, P., Keshwani, M.M., Oruganty, K., Nene, A.K., Kannan, N., Taylor, S.S., and Kornev, A.P. (2013). Deciphering the structural basis of eukaryotic protein kinase regulation. PLoS Biol 11, e1001680.
- Featured article Confirming the Importance of the R-Spine: New Insights into Protein Kinase Regulation. Richard Robinson. PLoS Biol 11(10): e1001681.
- Hu, J., Stites, E.C., Yu, H., Germino, E.A., Meharena, H.S., Stork, P.J., Kornev, A.P., Taylor, S.S., and Shaw, A.S. (2013). Allosteric activation of functionally asymmetric RAF kinase dimers. Cell 154, 1036-1046.
- Taylor, S.S., Shaw, A., Hu, J., Meharena, H.S., and Kornev, A. (2013). Pseudokinases from a structural perspective. Biochem Soc Trans 41, 981-986.
- Zan, J., Cicirelli, E.M., Mohamed, N.M., Sibhatu, H.M., Kroll, S., Choi, O., Uhlson, C.L., Wysoczynski, C.L., Murphy, R.C., Churchill, M.E., Hill, R.T., and Fuqua, C. (2012). A complex LuxR-LuxI type quorum sensing network in a roseobacterial marine sponge symbiont activates flagellar motility and inhibits biofilm formation. Molecular microbiology 85, 916-933.
- Churchill, M.E., Sibhatu, H.M., and Uhlson, C.L. (2011). Defining the structure and function of acyl-homoserine lactone autoinducers. Methods Mol Biol 692, 159-171.
- Tizzano, M., Gulbransen, B.D., Vandenbeuch, A., Clapp, T.R., Herman, J.P., Sibhatu, H.M., Churchill, M.E., Silver, W.L., Kinnamon, S.C., and Finger, T.E. (2010). Nasal chemosensory cells use bitter taste signaling to detect irritants and bacterial signals. Proc Natl Acad Sci USA 107, 3210-3215.
- Li, H., Das, A., Sibhatu, H.M., Jamal, J., Sligar, S.G., and Poulos, T.L. (2008). Exploring the electron transfer properties of neuronal nitric-oxide synthase by reversal of the FMN redox potential. J Biol Chem 283, 34762-34772.
Biography
Hiruy received his PhD in Biomedical Sciences from UC San Diego (UCSD). As a National Science Foundation (NSF) Graduate Research Fellow his research focused on understanding the biochemical and biophysical principles governing Protein Kinases to identify novel cancer therapeutics under the mentorship of Dr. Susan Taylor. He then completed his postdoctoral training at the Massachusetts Institute of Technology (MIT) under the mentorship of Dr. Li-Huei Tsai. As a UNCF/Merck, BWF PDEP, and Alana postdoctoral fellow his research focused on understanding the genetic, molecular and cellular mechanisms governing the neurodevelopmental abnormalities observed in Down syndrome.

